Chemical composition of the atmosphere
The atmosphere is made up of air (a homogeneous mixture of gases in different proportions), and atmospheric dust, small particles that float in the air (pollen, fine sand, volcanic ash, etc.).
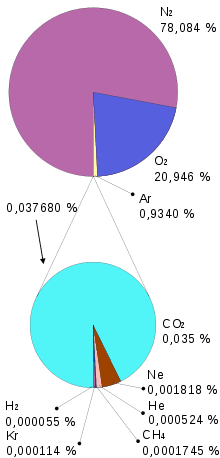
The composition of the air is:
- 78.08% Nitrogen. It is an inert gas that does not react with almost any substance. It is an inert gas.
- 20.95% Oxygen. It combines with other substances by oxidizing them. Needed for respiration. It is soluble in water. It is generated in photosynthesis (plants, algae and some bacteria).
- 0.93 Argon.
- 0.03% CO2. It is obtained from the combustion of fossil fuels and the respiration of living beings. Plants use it in photosynthesis.
- Other gases (O3, Neon, Hydrogen, Helium, Methane, Krypton, etc.).
- Water vapor. It comes from the evaporation of surface water and the transpiration of plants.
By Nicolás Lichtmaier [Public domain], via Wikimedia Commons
Air density decreases at high altitudes. Most of the air mass is in the low-lying areas, attracted by the force of Earth's gravity. On the other hand, in the higher areas, the air is thinner and lighter, with less density.
Air density and atmospheric pressure decrease height.